What Happens To Wax In A Fire?
Wax is defined as a substance made from oils or fatty acids that becomes thick and solid when cooled but melts at relatively low temperatures (Wax Definition & Meaning – Merriam-Webster). It is composed of long-chain hydrocarbons that can be either natural, such as beeswax produced by honey bees, or synthetic, like paraffin wax made from petroleum. When heated, solid wax transitions into a molten liquid state before vaporizing at higher temperatures.
The melting process of wax depends on its composition but generally occurs between 120-200°F. As heat is applied, the solid crystalline structure of the wax molecules becomes less organized and rigid. The hydrocarbons gain energy and begin sliding past each other more freely, causing the wax to liquefy. Melting points vary based on the length of the hydrocarbon chains, with longer chains requiring more energy to overcome the stronger intermolecular forces holding the molecules together in a solid state.
Composition of Wax
Wax is primarily composed of long-chain hydrocarbons, which are molecules made up of carbon and hydrogen atoms. The exact chemical composition can vary between different types of waxes. Some of the main ingredients in wax include saturated hydrocarbons, unsaturated hydrocarbons, free fatty acids, and wax esters (Buchholz, 2022).
One of the most common types of wax is paraffin wax, which consists mostly of straight chain n-alkanes. Beeswax contains some n-alkanes as well, but also has a significant proportion of wax esters and free fatty acids (Tulloch & Hoffman, 1972). Carnauba wax contains n-alkanes along with fatty acid esters, hydroxylated fatty acids, and fatty alcohols. The differences in chemical composition lead to variations in properties like melting point, hardness, and flexibility between the different wax types.
Wax esters, which are fatty acids linked to fatty alcohols, are a key component that distinguishes animal waxes like beeswax from plant and mineral waxes. Wax esters help make beeswax more pliable and fracture resistant compared to paraffin wax (Buchholz, 2022). The composition of wax has an important impact on its functional properties and applications.
References:
Buchholz, K. (2022). Wax in Biochemistry: Properties, Functions, and Contrasts of Different Types. Creative Proteomics. https://www.creative-proteomics.com/resource/waxes-biochemistry-functions-contrasts.htm
Tulloch, A. P., & Hoffman, L. L. (1972). Canadian Beeswax: Analytical Values and Composition of Hydrocarbons, Free Acids and Long Chain Esters. Journal of the American Oil Chemists Society, 49(12), 696–699. https://doi.org/10.1007/BF02635647
Melting Point
The melting point of wax refers to the temperature at which solid wax transitions to a liquid state. This temperature varies based on the type of wax. According to BellaOnline, paraffin wax, commonly used in candlemaking, has a melting point around 120 to 170°F depending on whether it is low, medium or high melt point paraffin wax.
As noted on Quora, typical paraffin wax melts somewhere around 135 to 145°F. Beeswax has a slightly higher melting point between 144 to 147°F. Other types of waxes like soy wax and palm wax melt at lower temperatures from 115 to 135°F.
The differences in melting points between waxes impact their usage. Low melt point waxes are desirable for container candles so the wax pools properly. Higher melt point waxes like beeswax are preferred for pillar candles and candles in warm environments.
Heating Wax
When wax is exposed to heat, the molecules begin to move and vibrate faster, causing intermolecular forces between them to weaken. This allows the rigid structure of solid wax to loosen and become more fluid, resulting in the melting of the wax into a liquid state. According to Waxing and Weather, the melting point of wax can range from 120 to 180 degrees Fahrenheit depending on the type of wax. Hard waxes, for example, melt between 120-130 degrees F.
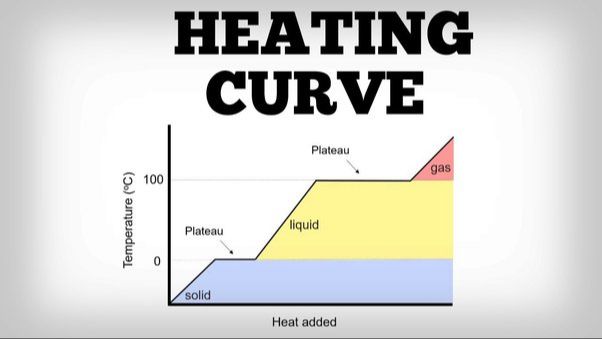
The rate at which wax melts depends on the intensity of the heat source and how quickly it can transfer thermal energy into the wax. A higher temperature heat source like the direct flame of a candle will melt wax faster than a lower temperature heat source like a radiator or the sun. The amount of wax present also plays a role. Melting a large block of wax requires more energy input and takes longer compared to melting a small amount of wax. This effect can be seen when melting wax for hair removal or candle making – smaller strips or portions melt quicker than larger sections. Overall, applying consistent heat accelerates the melting process by steadily adding energy to break down the wax’s solid structure.
Wax Burning
When a candle is lit, the flame melts the solid wax near the wick. The wick allows the melted wax to continue being drawn up so it can vaporize and keep the flame burning. As the flame melts more of the wax, a pool of liquid wax forms around the wick.
The candle wax melts because of the heat of the flame. The melting point of wax varies, but paraffin wax, a common candle wax, melts at about 130-150°F. When the wax reaches this temperature, it liquefies from a solid to a molten state that allows it to be drawn up the wick.
The heat of the candle flame can melt wax up to a half inch or more away from the wick. This forms the characteristic pool of liquid wax during burning. The depth and shape of the wax pool depends on factors like the type of wax, room temperature, candle diameter, and burn time.
Wax Vapors
When a candle burns, the heat from the flame melts the solid wax into a liquid. This liquid wax is then drawn up the wick through capillary action. At the flame, the liquid wax reaches high temperatures and vaporizes into a hot gas or vapor
According to Flinn Scientific, “As the wick burns, the heat melts the wax, and the liquid is “wicked up” where the heat of the flame vaporizes it. The wax vapor reacts with oxygen in the air, resulting in the flame.”[1] The vaporized wax molecules provide fuel that react with oxygen to keep the candle flame burning.
The vaporized wax, along with vaporized dyes and fragrances, rise up and are consumed in the flame. The production of wax vapors through melting and vaporization is crucial for sustaining the candle’s flame.
[1] https://www.flinnsci.com/wax-vapor-combustion-in-a-test-tube/dc91610/
Wax Residue
When wax burns, it leaves behind a residue known as soot. This burnt wax residue is made up of the impurities and additives that were present in the original wax before burning. As the wax melts and vaporizes, these non-volatile components are left behind as a black, carbon-rich solid residue [1]. This is similar to how burnt food in a pan leaves behind charred bits stuck to the bottom. The residue can build up over repeated burning and should be cleaned to avoid accumulation [2].
Wax Fire Safety

When burning wax, it’s important to take precautions to prevent wax fires. Wax has a low flash point, meaning it ignites easily. To prevent wax fires:
- Use caution when melting wax. Melted wax is flammable and can easily catch fire if overheated or spilled onto an open flame. Monitor melted wax carefully and keep away from open flames, sparks, and heat sources.
- Pour melted wax slowly and carefully. Spilled wax can ignite if it comes into contact with a flame or hot surface.
- Allow ample wax cooling time. Molten wax stays hot and should be allowed to fully harden before handling candles or other wax items. Rushing the cooling process can lead to spills and potential wax fires.
- Trim wicks to 1⁄4 inch before lighting candles. Long wicks cause candles to burn too hot, increasing the risk of flaring up and catching fire.
- Keep burning candles away from flammable materials like curtains, paper, fabrics. The heat from a candle flame can ignite nearby combustible materials.
- Don’t burn candles near open windows or drafts. Blowing air currents can cause candles to flare up unexpectedly.
- Extinguish candles when leaving a room or before going to sleep. Never leave burning candles unattended.
Following basic precautions when working with wax can help prevent accidental wax fires and make the experience safer and more enjoyable.
Other Uses for Melted Wax
Melted wax has many uses beyond just burning in candles or fires. Some of the most popular uses for melted wax are in arts, crafts, polishes, and waterproofing materials.
In arts and crafts, melted wax is commonly used to make candles, wax seals, batik fabrics, and encaustic paintings. The melted wax can be mixed with dyes or pigments and then applied to fabric or paper. As it cools and hardens, it creates interesting crackled textures and layers. Batik is an Indonesian technique that uses melted wax to resist dye on fabrics like silk or cotton. Encaustic painting also utilizes melted beeswax combined with pigments to create vivid works of art.

Melted wax can provide a protective waterproof coating for a variety of materials. For example, wax helps strengthen and weatherproof canvas tents and tarps. Rubbing melted beeswax on leather helps repel water and condition the material. Melted wax is also used to seal the cork stoppers of wine bottles.
In polishes and finishes, wax adds shine and luster. Melted beeswax is a key ingredient in products like furniture polish, wood finish, and shoe polish. The wax helps create a protective layer while buffing items to a lustrous shine. Wax polishes are common for finished wood surfaces like tables, floors, and musical instruments.
Overall, melted wax is a versatile material with many possibilities beyond just fueling a flame. Crafters, artists, and woodworkers are just a few of the people who find uses for melted wax in their work and hobbies.
(Source: https://www.mannlakeltd.com/education-crafts-gifts/wooden-solar-wax-melter/)
Conclusion
In summary, wax is composed of hydrocarbons that have a low melting point, allowing the wax to liquefy when exposed to heat. As wax is heated, it melts into a liquid and eventually vaporizes into a flammable gas. When burned, wax produces heat, light, carbon dioxide, water vapor, and some residual carbonaceous material. Understanding the properties of wax is crucial for utilizing it safely and efficiently in applications like candles, crayons, cosmetics, and art. Wax can be an extremely useful material, but caution must be exercised when melting or burning it to avoid accidental fires or injuries. This overview explored what happens on a chemical level when wax interacts with high temperatures and flames. The takeaway is that wax fundamentally changes states and chemical composition when burned, yielding both energy and byproducts. Being mindful of the science and safety precautions allows us to harness the unique benefits of wax.<(https://www.goodreads.com/en/book/show/23454794)>